Deregulation of the Ras-MAPK pathway is an early event in many different cancers and a key driver of resistance to targeted therapies. Kinase suppressor of Ras (KSR) is a MAPK scaffold. KSR exhibits allosteric regulation through dimerization with RAF. In this study, researchers developed a class of compounds that stabilize the inactive state of KSR. They identify APS-2-79 as a small-molecule mimic of KSR alleles that suppress oncogenic Ras mutations.
APS-2-79 modulates KSR-dependent MAPK signalling by antagonizing RAF heterodimerization as well as the conformational changes required for phosphorylation and activation of KSR-bound MEK. APS-2-79 increases the potency of several MEK inhibitors specifically within Ras-mutant cell lines by antagonizing the release of negative feedback signalling, demonstrating the potential of targeting KSR to improve the efficacy of current MAPK inhibitors. Moreover, APS-2-79 (5 μM) also suppresses KSR-stimulated MEK and ERK phosphorylation.
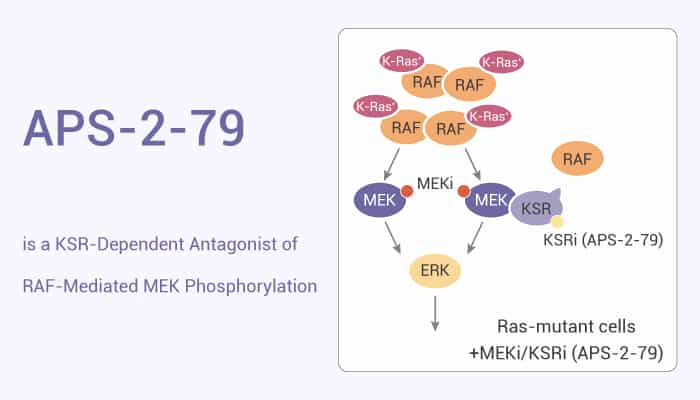
Indeed, APS-2-79 lacks direct activity against the highly homologous active RAF family kinases, including recombinant BRAF and CRAF, or cellular BRAF(V600E). APS-2-79 functions as an antagonist of MEK phosphorylation by RAF through direct binding of the KSR active site. In addition, APS-2-79 binds directly to KSR2 within the KSR2-MEK1 complex. APS-2-79 (1 μM ) enhances the efficacy of the clinical MEK inhibitor Trametinib within cancer cell lines containing K-Ras mutations. APS-2-79 (1 μM ) hinders RAF-mediated MEK phosphorylation in a KSR-dependent manner. Furthermore, the small molecule APS-2-79 (5 μM) mimics KSR alleles that suppress oncogenic Ras mutations
All in all, KSR acts as a druggable regulator of oncogenic Ras, and further suggests co-targeting of enzymatic and scaffolding activities within Ras–MAPK signalling complexes as a therapeutic strategy for overcoming Ras-driven cancers.