Protein Phosphatase 2A (PP2A) is a serine/threonine phosphatase. It regulates multiple signaling cascades implicated in cancer progression, including downstream effectors of KRAS. Inhibition of PP2A contributes to oncogenesis in multiple tumor types, highlighting the importance of this protein in maintaining normal kinase activity. Pancreatic ductal adenocarcinoma (PDA) cells have reduced PP2A activity and an upregulation of the PP2A inhibitors, CIP2A and SET. Further, high CIP2A expression in PDA patients correlates with decreased overall survival. Suppression of PP2A may significantly contribute to PDA cell survival. As such, compounds that activate PP2A are emerging as promising cancer therapeutics. The majority of PP2A activating agents disrupt the interaction between PP2A and CIP2A or SET, indirectly increasing PP2A activation and reducing tumor growth. In this study, SMAPs directly bind the PP2A A subunit and predominately function through PP2A activation. SMAP-2 (DT-1154) is an orally active PP2A activator, with anti-cancer activity.
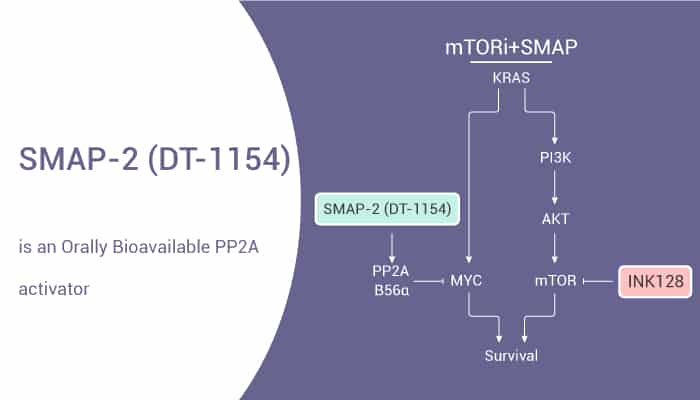
Activation of PP2A and inhibition of mTOR synergistically reduce MYC signaling and decrease tumor growth in pancreatic ductal adenocarcinoma. SMAP-2 reduces cell viability of pancreatic ductal adenocarcinoma (PDA) cell lines in a dose-dependent manner. The combination of SMAP-2 and INK128 results in a significant loss of both MYC and AKT/mTOR signaling pathway. SMAP-2 also significantly reduces abdominal aortic aneurysms (AAA) incidence and aortic dilation in pro-AAA apolipoprotein E-null (ApoE-/-) mice. Moreover, SMAP-2 has anti-tumor activity in the pancreatic cancer mice model.
In summary, SMAP-2 effectively reduces MYC levels in HPAFII and PANC89 cells. SMAP-2 combined with INK128 has a greater suppression of MYC in these cell lines. Thus, the use of PP2A-activating compounds in combination with kinase inhibitors is a novel therapeutic strategy in PDA.
Reference:
Brittany L Allen-Petersen, et al. Cancer Res. 2019 Jan 1;79(1):209-219.