Phosphoinositide 3-kinases (PI3Ks), also called phosphatidylinositol 3-kinases, are a family of enzymes involved in cellular functions. Class I PI3K enzymes consist of four distinct catalytic isoforms, PI3Kα, PI3Kβ, PI3Kδ and PI3Kγ. There are three major classes of PI3K enzymes, being class IA widely associated to cancer. Class IA PI3K, heterodimeric lipid kinases composed of a catalytic subunit (p110α, p110β, or p110δ; encoded by PIK3CA, PIK3CB, and PIK3CD genes, respectively) and a regulatory subunit (p85). The PI3K pathway plays an important role in many biological processes, including cell cycle progression, cell growth, survival, actin rearrangement and migration, and intracellular vesicular transport.
Wortmannin (also known as SL-2052 or KY-12420), a fungal metabolite, is a potent, selective, irreversible, and orally active PI3K inhibitor. However, Wortmannin potently inhibits Plk1 in addition to the PI 3-kinase superfamily. It covalently labels PI1K and PI3K by targeting conserved lysine residues in their ATP binding sites. In particular, Wortmannin affects the downstream targets of PI3K. For example, this compound reduces the phosphorylation of p53 on serine 20 induced by DNA damage. In addition, Wortmannin can affect multiple functions of PI3K in cell cycle progression and at the DNA damage checkpoint. Furthermore, Wortmannin is also an autophagy inhibitor and can inhibit the encystation of Acanthamoeba cells. Besides, Wortmannin inhibits the growth of murine C3H and human MCF-7 mammary tumors in vivo.
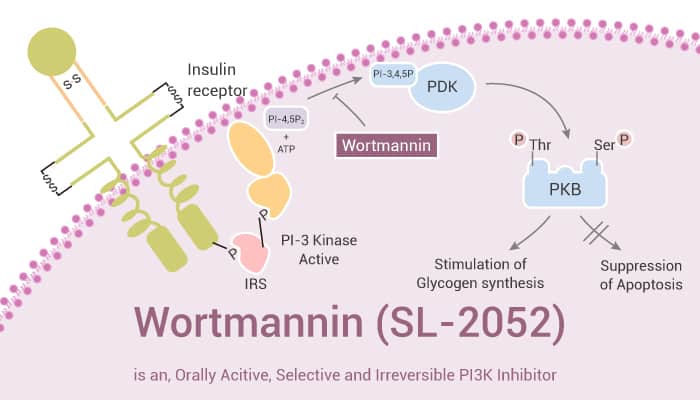
Taken together, Wortmannin is a potent, selective, irreversible, and orally active PI3K inhibitor, with anticancer effects.
References:
[1] Eun-Kyung Moon, et al. Antimicrob Agents Chemother. 2015 Jul;59(7):4020-5.
[2] Yongsheng Liu, et al. J Biol Chem. 2007 Jan 26;282(4):2505-11.