Polatuzumab vedotin is an antibody-drug conjugate targeting CD79b for Large B-cell lymphomas (LBCL) Research. CD79b is a surface antigen whose expression is restricted to pre‐B and mature B cells. Besides, CD79b is expressed on nearly all major subtypes of B-cell-derived NHL. While, Large B-cell lymphomas (LBCL) is a fast-growing type of non-Hodgkin’s lymphoma (NHL), which is cancer that develops in the lymphatic system and affects B-cell lymphocytes.
Moreover, CD79b functions as a signaling component of the B-cell receptor and is highly expressed in most types of B-cell malignancies including DLBCL. In addition, Research shows that antibodies that bind to CD79b rapidly internalize and traffic to the lysosomal compartment, making CD79b a suitable tumor antigen for the targeted delivery of cytotoxic agents.
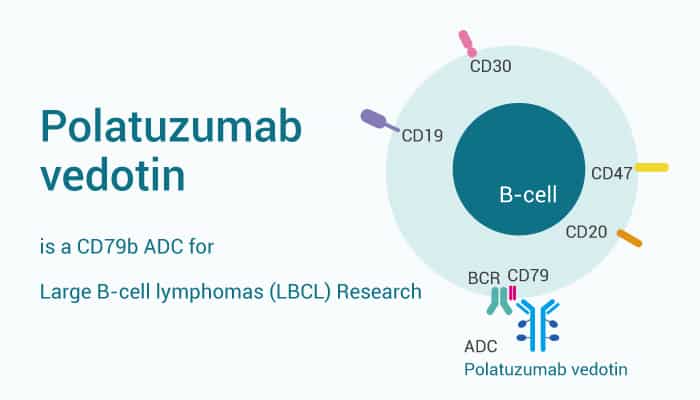
Polatuzumab vedotin is an antibody-drug conjugate (ADC) for non‐Hodgkin’s lymphoma. Importantly, It contains a humanized anti-CD79b IgG1 monoclonal antibody linked to monomethyl auristatin E (MMAE), an anti-mitotic agent. Particularly, Polatuzumab vedotin binds human CD79b, but it does not bind CD79b of most non-clinical species.
Polatuzumab vedoti shows high binding affinity to human CD79b with a KD value of 1.83 nM. Besides, Polatuzumab vedotin also shows high binding activity to FcγRIA. Meanwhile, Polatuzumab vedoti (1, 2 mg/kg; i.v.; once) significantly inhibits tumor growth in a dose-dependent manner in BJAB‐PD.cyCD79b.E3 xenograft tumors. Furthermore, Polatuzumab vedotin (0.3, 1, 3, 6, 12 mg/kg; i.v.; once) shows anti-tumor activity in a mouse tumor xenograft model of human diffuse large B-cell lymphoma WSU-DLCL2. More than that, Polatuzumab vedoti (1, 3, 5 mg/kg; i.v.; every 3 weeks for 140 days) does not show any decrease in peripheral CD20+ B cells in cynomolgus monkeys.
All in all, Polatuzumab vedotin is an antibody-drug conjugate targeting CD79b and has the potential for the research of Large B-cell lymphomas (LBCL).
Reference:
[1] Liebers N, et al. Blood Adv. 2021 Jul 13;5(13):2707-2716.
[2] Li D, et al. Br J Pharmacol. 2019 Oct;176(19):3805-3818.