Isocitrate dehydrogenase (IDH) is an enzyme that catalyzes the oxidative decarboxylation of isocitrate. Firstly, IDH exists in three isoforms in humans: IDH3 catalyzes the citric acid cycle while converting NAD+ to NADH in the mitochondria. Secondly, IDH1 and IDH2 isomers catalyze the same reaction outside the context of the citric acid cycle. Then they use NADP+ as a cofactor instead of NAD+. Moreover, IDH1 is located in the cytoplasm and peroxisomes. At the same time, IDH2 and IDH3 are located in the mitochondria. Importantly, the study of isocitrate dehydrogenase (IDH) in cancer is divided into wild-type IDH and mutant IDH. IDH1 mutation status is currently included as an essential criterion for the classification of gliomas in the World Health Organization Classification of Tumors of the Central Nervous System. Then, a number of small molecule inhibitors have been developed to specifically inhibit mutant forms of IDH.
Ivosidenib (AG-120) is an orally active mIDH1 inhibitor for AML research.
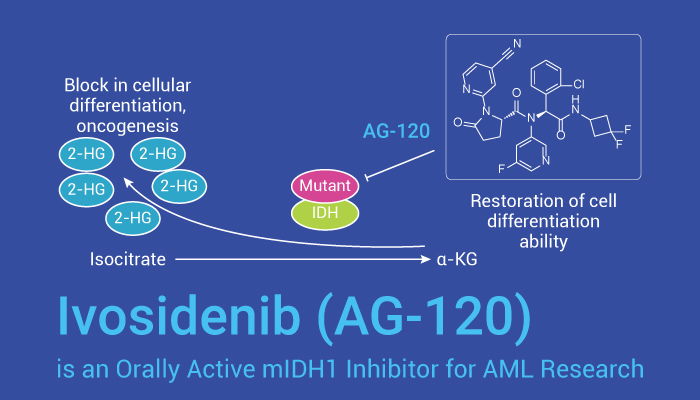
Ivosidenib (AG-120) is an orally active inhibitor of the isocitrate dehydrogenase 1 mutant (mIDH1) enzyme. Also, it exhibits profound d-2-hydroxyglutatrate (2-HG) lowering in vivo. Besides, it has the potential for AML therapy due to its acceptable safety profile and clinical activity. In some in vitro studies, Ivosidenib (AG-120) (0-13 μM; 48 hours) inhibits several IDH1-R132 mutants with potency similar IC50 values: IDH1-R132H (IC50=12 nM); IDH1-R132C (IC50=13 nM); IDH1-R132G (IC50=8 nM); IDH1-R132L (IC50=13 nM); IDH1-R132S (IC50=12 nM), respectively. In addition, AG-120 (p.o., 50 mg/kg and 150 mg/kg) declines tumor 2-HG concentration rapidly. For more, it results in maximum inhibition (92.0% and 95.2% at the 50 mg/kg and 150 mg/kg doses, respectively) achieved at -12 h post-dose.
In conclusion, Ivosidenib (AG-120) is an orally active inhibitor of mIDH1 with a favorable safety profile and clinical activity. Besides, it has the potential for AML research.
References:
[1] Janeta Popovici-Muller, et al. ACS Med Chem Lett. 2018 Jan 19;9(4):300-305.
[2] Daniel J. Urban, et al. Sci Rep. 2017 Oct 6;7(1):12758.