A study from Andrew P. Turnbull reported two compounds, FT671 and FT827, as USP7 (ubiquitin-specific protease 7) inhibitors.
Ubiquitination controls the stability of most cellular proteins, and its deregulation can lead to human diseases, including cancer. Deubiquitinases remove ubiquitin from proteins, and their inhibitory effects can induce degradation of specific proteins, possibly including other non-pharmacological targets. For instance, inhibition of USP7 leads to the degradation of the oncogenic E3 ligase MDM2 and the reactivation of p53 (tumor suppressor) in various cancers.
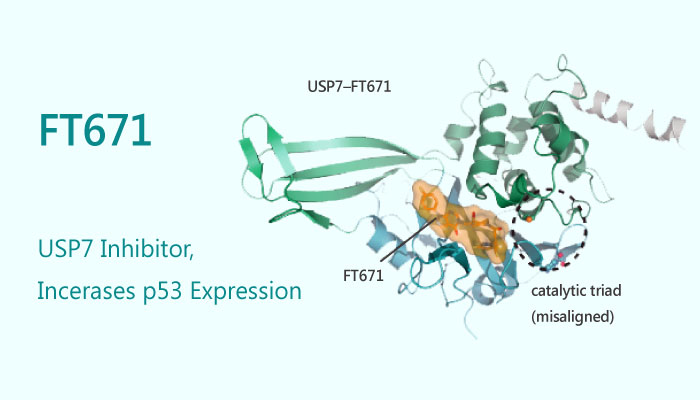
In the study, FT671, identified as the USP7 inhibitor, binds to the USP7 catalytic domain (USP7CD; residues 208-560). Additionally, it destabilizes USP7 substrates including MDM2, increases levels of p53, and results in the transcription of p53 target genes, induction of the tumour suppressor p21. Besides, the Kd value of FT671 was 65 nM for binding to the USP7 catalytic domain and the IC50 was 52 nM for USP7CD.
In vitro, in HCT116 or bone osteosarcoma (U2OS) cells, FT671 increased p53 protein levels, leading to induction of p53 target genes including BBC3, CDKN1A (p21) and MDM2. Additionally, it also lead to the degradation of N-Myc and upregulation of p53 in the neuroblastoma cell line IMR-32. FT671 also stabilized p53 in the MM.1S multiple myeloma cell line.
Moreover in vivo, FT671 treatment with the dosage of 100 mg/kg and 200 mg/kg by oral gavage, daily) led to a significant dose-dependent inhibition of tumor growth in MM.1S xenograft mouse model in which non-obese diabetic-severe combined immunodeficient (NOD-SCID) mice implanted with MM.1S cells. And this compound was well-torelated in treated mice even with high dosage.
Overall, FT671 is a promising candidate drug to treat cancers like multiple myeloma.