Chromosome region maintenance 1 (CRM1) is originally identified in yeast. It is essential for maintaining chromosome structure. CRM1 protein is a nuclear export receptor belonging to the karyopherin β family of transport receptors. CRM1 binds to different cargo proteins bearing leucine-rich nuclear export sequences leading to nuclear export of cargo proteins and mRNAs. Exported cargo proteins include tumor suppressors, such as p53, BRCA1, survivin, nucleophosmin, and adenomatous polyposis coli that shuttle between the nucleus and cytoplasm. Various malignancies, such as breast, cervical, ovarian, and pancreatic cancers, glioma, and osteosarcoma, have overexpressed CRM1. Knockdown of CRM1 protein via siRNA causes cell growth inhibition and apoptosis in cancer cells. However, CRM1 downregulation unaffected normal tissues. Thus, CRM1-mediates nuclear export is a potential target for treating cancers.
CBS9106, a synthetic small-molecular inhibitor of CRM1, decreases CRM1 protein through proteasomal degradation without affecting CRM1 mRNA levels.
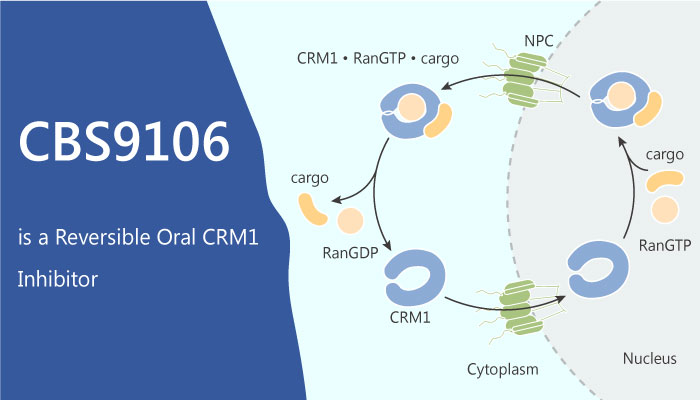
In this study, CBS9106 inhibits CRM1-dependent nuclear export. Furthermore, CBS9106 causes arrest of the cell cycle and induces apoptosis in a time- and dose-dependent manner. Meanwhile, CBS9106 can inhibit a broad spectrum of cancer cells, including multiple myeloma cells. CBS9106 reduces CRM1 protein levels significantly without affecting CRM1 mRNA expression. However, adding bortezomib or LMB could reverse this effect. Moreover, CBS9106 reacts with a synthetic CRM1 peptide that contains Cys528 but not with a Cys528 mutant peptide. On the other hand, oral administration of CBS9106 significantly suppresses tumor growth and prolongs survival in mice. Those mice bearing human multiple myeloma xenograft do not show a significant loss in body weight. A reduced level of CRM1 protein is also observed in tumor xenografts isolated from mice treated with CBS9106.
Taken together, CBS9106 is a novel reversible CRM1 inhibitor and a promising clinical candidate.
Reference: