Multiple myeloma represents a heterogeneous disease with various oncogenic mutations, chromosomal translocations, and copy number variations. The mitotic kinase T-LAK cell‐originated protein kinase/PDZ‐binding kinase ( TOPK/PBK) associates with the proliferation of tumor cells, maintenance of cancer stem cells, and poor patient prognosis in many cancers. In addition, TOPK plays an indispensable role in the mitosis of cancer cells. In this report, researchers establish high-level TOPK expression in multiple myeloma and show for the first time that TOPK inhibition effectively kills multiple myeloma cells. Especially, OTS514 is a highly potent TOPK inhibitor. In particular, OTS514 inhibits TOPK kinase activity with a median IC50 value of 2.6 nM.
OTS514 induces cell cycle arrest and apoptosis at nanomolar concentrations in a series of human myeloma cell lines and prevents outgrowth of a putative CD138+ stem cell population.
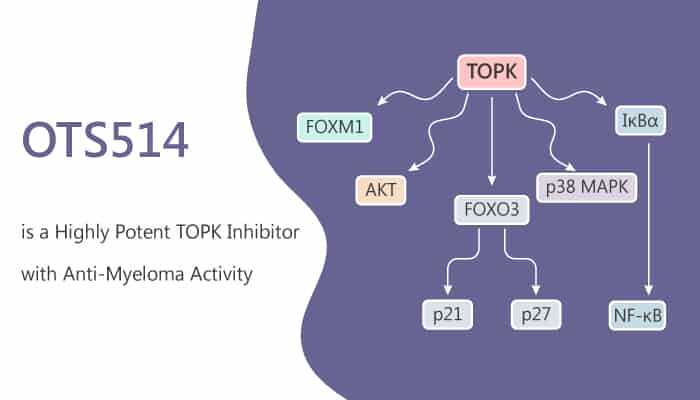
Moreover, it activates FOXO3 and CDK-inhibitory proteins while disrupting pro-survival kinase cascades. In bone marrow cells from multiple myeloma patients, OTS514 treatment exhibits preferential killing of the malignant CD138+ plasma cells compared with the CD138−compartment. Furthermore, it strongly suppresses the growth of TOPK-positive cancer cells.
In an aggressive mouse xenograft model, OTS964 (100 mg/kg; orally; 5 days per week) reduces tumor size by 48%‐81% compared to control. Particularly, TOPK inhibition also induces loss of FOXM1 and disrupts AKT, p38 MAPK, and NF‐κB signaling. The effects of OTS514 are independent of p53 mutation or deletion status. The combination of OTS514 and Lenalidomide leads to an additive loss of antioxidative capacity. OTS514 activates a p21/p27 response irrespective of p53 status. OTS514 treatment causes a marked decrease in the phosphorylated form of IκBα.
All in all, OTS514 is a potent TOPK inhibitor with anti‐myeloma activity.