CDK6 plays a significant role in cell cycle entrance. However, overexpression of CDK6 correlates to CDK4/6 inhibitor resistance in breast cancer cell lines and patient samples. Moreover, point mutation of CDK6 could possibly result in attenuation of drug binding affinity or hyperactivation of CDK6. Thus, there is an urgent need to develop a practical strategy against CDK6-centered malignancy. PROTAC is an emerging chemical biology approach for targeted protein depletion by exploiting the intracellular ubiquitin–proteasome system. A typical PROTAC molecule is bifunctional by combining a target-selective ligand and a specific E3 ligase recruiting ligand via a linker. The resulting PROTAC could thus recruit the E3 ligase onto target protein and induce the ubiquitination and protein degradation via the proteasome. CP-10 is a PROTAC with specific and remarkable CDK6 degradation. It has a DC50 of 2.1 nM.
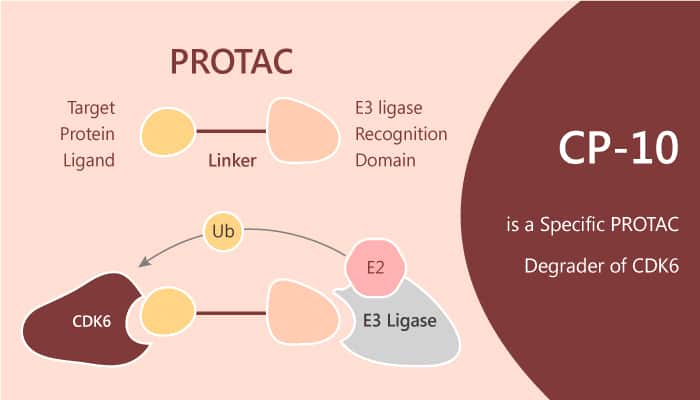
CP-10 is a PROTAC with highly selective, specific, and remarkable CDK6 degradation.
CP-10 links CDK6 inhibitor Palbociclib and E3 ligase CRBN recruiter pomalidomide. It significantly induces degradation of CDK6 with a DC50 of 2.1 nM. In this study, CP-10 with the conjugation of palbociclib and pomalidomide demonstrates the best degradation efficacy. Firstly, CP-10 induces nearly 72% degradation of CDK6 at 10 nM and 89% at 100 nM in human glioblastoma U251 cells. The degradation of CDK4 induced by CP-10 is far weaker than that of CDK6. Thus, CP-10 is highly selective for CDK6 without significant off-target effect. Secondly, CP-10 displays a good cell inhibition potential (IC50≈10 nM) in multiple myeloma cell MM.1S and mantle cell lymphoma cells (in Mino, IC50≈8 nM) or comparable activities in leukemia cells.
In summary, CP-10 could inhibit proliferation of several hematopoietic cancer cells with impressive potency including multiple myeloma. Moreover, CP-10 can still degrade mutated and overexpressed CDK6. Thus, it suggests the growing trends of potential clinical benefits of PROTAC techniques.
Reference:
Su S, et al. J Med Chem. 2019 Aug 2.