Lysyl oxidase (LOX) is a copper and quinone-cofactor containing amine oxidase. It is an inactive pro-enzyme and can be cleaved to a mature active form by extracellular metalloproteases. LOX prompts the formation of covalent bonds in collagens and elastin. Thereby, it increases the stiffness of the ECM.
Besides, many cancers, such as colon, breast, prostate and lung cancers express high LOX expression. Moreover, LOX-mediated ECM stiffening leads to integrin activation and tumor initiation in a mouse model of breast cancer. It also drives the growth of the primary tumors in colorectal cancer. In this article, we will introduce how LOX regulates intracellular signaling in the tumor. An inhibitor, CCT365623 suppresses EGFR (pY1068) and AKT phosphorylation driven by EGF.
The property of an orally active LOX inhibitor, CCT365623 (IC50=0.89 μM) will be described in vitro and in vivo.
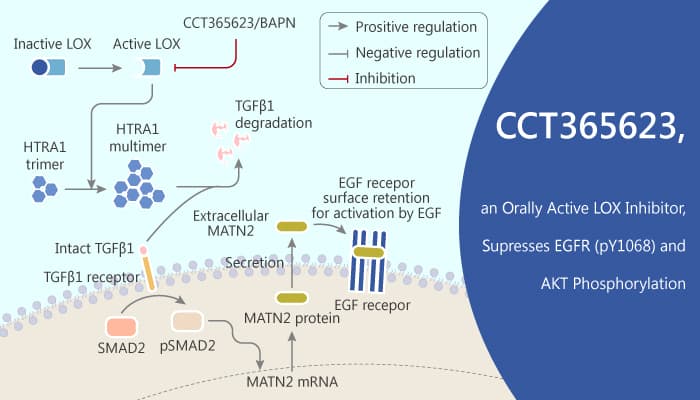
Firstly, CCT365623 inhibits LOX at 5 μM in the biosensor system and concentration-dependently decreased the protein levels of surface EGFR. In MDA-MB-231 cells, CCT365623 Significantly decreases the protein levels of pY1068 EGFR, pAKT, and MATN2. besides, CCT365623 disrupts HTRA1 multimerization, activates TGFβ1 signaling.
Nextly, in vivo, CCT365623 displays good stability in mouse liver microsomes and does not inhibit the cardiac potassium channel hERG. This result shows that CCT365623 exhibits extremely well-tolerance and has good pharmacokinetic properties.
Lastly, researchers exert a mouse model of spontaneous breast cancer that metastasizes to the lungs. CCT365623 at 70 mg/kg by oral significantly delays the development of the primary tumors. It exhibits a T1/2PO of 0.6 h and F% (oral bioavailability) of 45%. At the same time, it suppresses metastatic lung burden in the animals. Whatmore, the compound disrupts EGFR cell surface retention and delays the growth of primary and metastatic tumor cells.
In summary, CCT365623 as a potent LOX inhibitor suppresses EGFR (pY1068) and AKT phosphorylation driven by EGF. It has a great therapeutic promise that is also valuable for studying the LOX mechanism in vitro and in vivo.
Reference:
Tang H, et al. Nat Commun. 2019 Jul 18;10(1):3151.