Proteolytic targeting chimera (PROTAC) is a heterobifunctional small molecule that acts by inducing selective intracellular proteolysis. Specifically, bifunctional PROTAC molecule is including ligands of target protein (POI) and covalent ligands of E3 ubiquitin ligase (E3). After binding to POI, PROTAC can recruit E3 for POI ubiquitination. This ubiquitination is subject to proteasome-mediated degradation. Besides, PROTAC only needs to bind their targets with high selectivity, rather than inhibit the enzyme activity of the target protein. Moreover, PROTAC complements nucleic acid-based gene knockout/knockout techniques for targeting protein reduction. Furthermore, PROTAC can simulate pharmacological protein inhibition. PROTAC technology can be used to target a variety of proteins, including transcription factors, skeletal proteins, enzymes, and regulatory proteins.
Meanwhile, bromine domain and extra terminal (BET) proteins are “readers” of histone acetylation markers and play a role in gene transcription. The bet protein family consists of four conserved mammalian members (BRD2, BRD3, Brd4, and BRDT). Nonetheless, they regulate the expression of many immune-related genes and pathways. In response to infection and aseptic inflammation, abnormally expressed or dysfunctional bet is relevant to the activation of the pattern recognition receptor pathway. Importantly, this links the chromatin mechanism to innate immunity under disease or pathological conditions. Here, we will introduce a PROTAC based BET family degrade, SIM1.
SIM1 is a PROTAC Based BET Family Degrade.
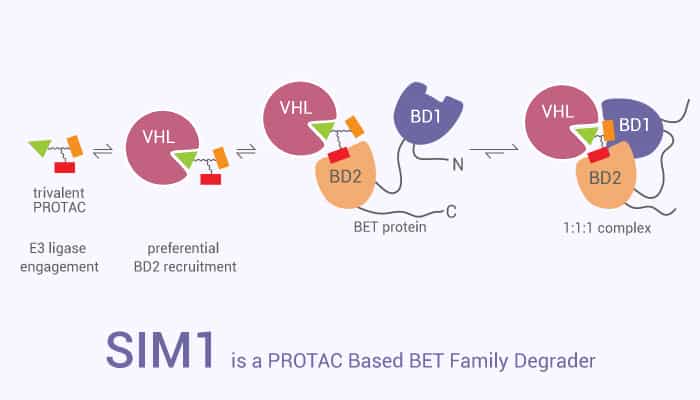
How does SIM1 work on the target? Let’s study it together. In the beginning, SIM1 is a potent von Hippel-Lindau (VHL)-based trivalent PROTAC. Particularly, SIM1 is capable of degradation for all BET family members, with a preference for BRD2 degradation (IC50=1.1 nM; Kd=186 nM). Interestingly, SIM1 shows sustained anti-cancer activity.
In the second place, SIM1 with 1 nM for 30 hours results in MV4-11 cells death after 6 hours. Obviously, SIM1degrades BET proteins in HEK293 cells. SIM1 induces conformational changes upon binding to the BET protein. SIM1 simultaneously engages with high avidity in both its bromodomains in a cis intramolecular fashion. Additionally, SIM1 engages BD1 and BD2 intramolecularly and forms a 1:1:1 ternary complex with VHL and BRD4.
All in all, SIM1 is a PROTAC Based BET Family Degrade.
References:
Satomi Imaide, et al. Biological and Medicinal Chemistry.