Ubiquitination is a post-translational modification where the polypeptide ubiquitin is attached to lysine residues of substrate proteins. This process is reversible, and ubiquitin removal is orchestrated by a large family of deubiquitinating enzymes (DUBs). DUBs fall into six subfamilies based on sequence and domain similarity. Ubiquitination and deubiquitination together with proteasomal activity control many aspects of cell physiology. Many of the enzymes involved have a relation with human disorders, including cancer. PR-619 is a broad-spectrum DUB inhibitor that nonetheless does not target other cysteine proteases such as cathepsin B or calpain 1. It also attenuates both the proliferation and migration of the mesothelioma cell line. PR-619 also inhibits the proliferation of the lung cancer cell line. It also shows EC50s ranging from 3.93 to 8.61 μM for USP4, USP8, USP7, USP2, and USP5, respectively. It induces ER stress and ER-stress related apoptosis.
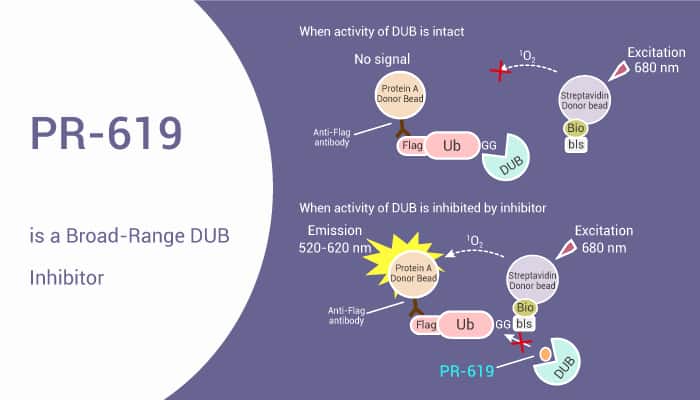
PR-619, a broad-spectrum DUBs inhibitor, is a tool to investigate the role of ubiquitination in various cellular processes.
PR-619 affects the microtubule network and led to the accumulation of small punctuated tau deposits around. It causes the dephosphorylation of tau. PR-619 also causes an increase in the abundance of ubiquitinated proteins within 24 h. Moreover, It leads to the induction of heat shock proteins and to an increase of ubiquitinated proteins. PR-619 affects the organization of the microtubule network in OLN-t40 cells. Furthermore, PR-619 induces ER Stress and ER-Stress related apoptosis on T24 and BFTC-905 cells. It induces polyubiquitination, Bcl-2 downregulation, and concurrent PARP cleavage in a dose-dependent manner. It also induces G0/G1 arrest in UC cells. In addition, PR-619 enhances the antitumor effect of Cisplatin on a Cisplatin-Naïve and Cisplatin-resistant UC Xenograft of nude mice.
In summary, PR-619 is a broad-range and reversible DUB inhibitor and induces ER stress and ER-stress related apoptosis.
Reference:
Mirzapoiazova T, et al. Int J Oncol. 2020 Jul;57(1):80-86.;
Altun M, et al. Chem Biol. 2011 Nov 23;18(11):1401-12.;
Kuan-Lin Kuo, et al. Cells. 2019 Oct 17;8(10):1268.