Mutant EGFRs are one of the major drivers of non-small-cell lung cancer (NSCLC). While reversible first-generation EGFR inhibitors are initially an effective treatment. However, a drug-resistant gatekeeper T790M mutation accounts for ∼50% of disease progression in patients with first-generation agents. Irreversible second-generation EGFR inhibitors are more potent against the T790M mutants. They covalently modify C797 in the ATP binding site of EGFR. However, these agents are dose limited by their exceptional potency against wild-type (WT) EGFR, which drives mechanism-based toxicity. It is important to overcome this limitation. It hopes the emergence of irreversible third-generation EGFR inhibitors that provide effective potency against both drug-resistant T790M mutants and the original activating mutants while sparing activity against WT EGFR. Mavelertinib is a selective, orally available, and irreversible EGFR inhibitor. The IC50s are 5-12 nM for Del, L858R, and double mutants T790M/L858R and T790M/Del, respectively.
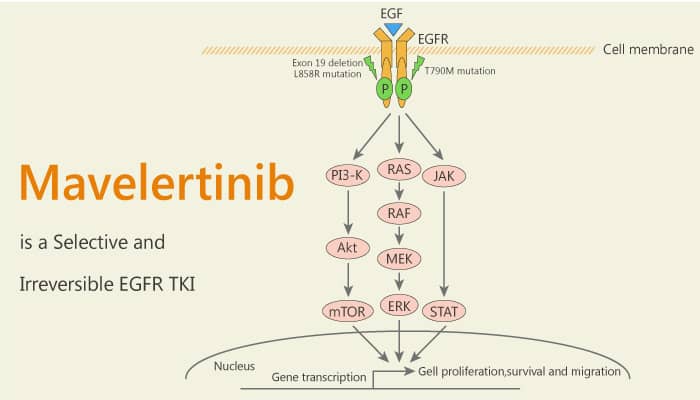
Mavelertinib is a high-affinity irreversible inhibitor targeting oncogenic EGFR mutants with selectivity over wild-type EGFR.
Mavelertinib also exhibits selectivity over wild-type EGFR (IC50=307 nM). It (10 μM) exhibits less than 50% effect or inhibition against all non-kinase targets. Moreover, it inhibits the hERG26 current with an IC50 > 100 μM. In addition, Mavelertinib exhibits low to moderate oral bioavailability (mouse 60%, rat 11%, dog 66%) following oral administration (mouse 1, rat 30, dog 3 mg/kg). It also exhibits short plasma half-lives (mouse 0.56, rat 0.28, dog 1.3 h) due to moderate to high plasma clearance (mouse 53, rat 49, dog 12 mL/min/kg) and low steady-state volume of distribution (mouse 1.48, rat 0.66, dog 0.94 L/kg) following intravenous administration (1 mg/kg to mouse, rat and dog).
In summary, Mavelertinib provides potent EGFR activity against the four common mutants (exon 19 deletion (Del), L858R, and double mutants T790M/L858R and T790M/Del), selectivity over wild-type EGFR, and desirable ADME properties. It has the potential for mutant EGFR-driven NSCLC research.
Reference:
Planken S, et, al. J Med Chem. 2017 Apr 13;60(7):3002-3019.